Photocatalysts are useful materials, with myriad environmental and energy applications, including air purification, water treatment, self-cleaning surfaces, pollution-fighting paints and coatings, hydrogen production and CO2 conversion to sustainable fuels.
An efficient photocatalyst converts light energy into chemical energy and provides this energy to a reacting substance, to help chemical reactions occur.
One of the most useful such materials is knows as titanium oxide or titania, much sought after for its stability, effectiveness as a photocatalyst and non-toxicity to humans and other biological organisms.
In new research appearing in the Journal of Physical Chemistry Letters, Scott Sayres and his research group describe their investigations into the molecular dynamics of titania clusters.
Such research is a basic step toward the development of more efficient photocatalysts.
The key to such advances is the ability to extend the time that electrons within the material persist in an excited state, as this fleeting duration is when titania can act as an efficient photocatalyst.

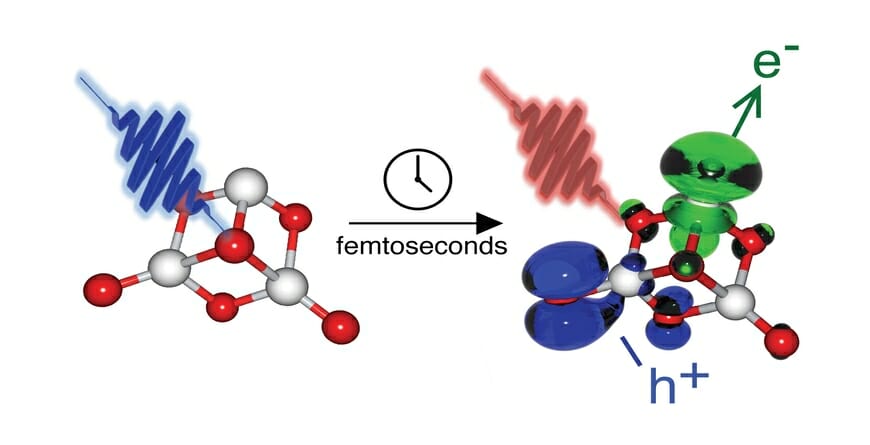
Probing the behavior of a photocatalyst in fine detail, however, is a tricky endeavor. The clusters are a nanometer or less in size (or 1/100,000th the width of a human hair) and the movements of electrons within the molecules under study take place on astonishingly brief time scales, measured in femtoseconds (or one millionth of a billionth of a second).
The new study explores neutral (uncharged) clusters of titania for the first time, tracking the subtle movements of energy using a femtosecond laser and a technique known as pump-probe spectroscopy. “We treat our lasers like cameras,” Sayres says. “We take pictures of where the energy is flowing over time.”
Sayres, a researcher in the Biodesign Center for Applied Structural Discovery, describes the significance of the current study:
“We've examined the smallest possible building blocks of titania to understand the relationship of how small changes in the material’s atomic structure influences the excited state lifetimes and flow of energy. Learning about how this happens can help redesign better photocatalysts in the future.”
The research will grace the supplementary cover of this publication, which is the premier journal for physical chemistry.